Impact Program
Grow your project to maximize its impact
At CIMTI we offer the Impact Program, an acceleration program for innovative projects in the health and social field aimed at projects in phase 3-5 of the Health Innovation Cycle.
The projects that are part of the Impact Program receive support in the conceptualization stages of the project, in the arrival to the market and in the implementation in the health and social system following our own methodology, the Health Innovation Cycle. Our methodology has been adapted from CIMIT Boston’s Healthcare Innovation Cycle which, with more than 30 years of experience in healthcare innovation, ensures efficient development of solutions and reduces the risk of failure.
The Impact Program, with a duration of 15 months, is estimated to help project teams advance between 1 and 2 phases of the Health Innovation Cycle.
To access the Impact Program, it must be done through the CIMTI Innovation Call or CIMTI Challenge Call.
Which services do we offer your project within the Impact Program?
The services offered by CIMTI aim at helping the projects reaching the market and becoming a standard of care.
CIMTI offers different services related to the 4 domains of the Health Innovation Cycle: technology, regulatory, market and business, and clinical; as well as other services related to financing, communication, training, access to CIMIT Boston, mentoring by the CIMTI team or access to the Catalan health ecosystem.
In total, the services offered in the Impact Program costs 85,000 euros.

Technology
Technology
- Advice sessions with experts
- Planification and execution of proof-of-concept tests and prototyping
- Access to manufacturing partners

Regulatory
Regulatory
- Advice sessions with experts
- Regulatory classification and pathway
- Review of instructions for use

Market and business
Market and business
- Description of the map of stakeholders
- Description of the value proposition
- Characterization of competing solutions
- Advice on the design and protection of the business model

Clinical
Clinical
- Access to clinical experts
- Access to experts to build the Advisory Board
- Access to stakeholders for the validation of the unmet need and the clinical workflow

Financing
Financing
- Support in the search for public funding
- Access to experts on private funding
- Advice to prepare investment rounds
- Review of investment proposals and partnership agreements

Communication
Communication
- Advice on online communication
- Advice on brand identity, logotype and corporate identity manual
- Project’s appearance in traditional media
- Promotional video of the project

Training
Training
- Optional access to all the training sessions organized by CIMTI

Access to the CIMIT of Boston
Access to the CIMIT of Boston
- Work session to define the business model and explore opportunities to access the American market

Mentoring by the CIMTI team
Mentoring by the CIMTI team
- Individualized advice and follow-up throughout the duration of the program
- Access to a network of mentors from the CIMTI community

Access to the Catalan health ecosystem
Access to the Catalan health ecosystem
- Advice with experts from public institutions in the health sector in Catalonia
CIMTI's Methodology: Healtcare Innovatioin Cycle
The process from the identification of an unmet need to the development of an innovative solution that becomes a standard of care is long and involves many challenges. For this reason, many of the innovative teams have difficulties during this process and even end up failing. The chances of carrying out this process successfully increase when teams have the necessary skills and experience to anticipate and overcome challenges. However, experience often comes from trial and error, which is an inefficient process.
For this reason, Boston’s CIMIT, based on the experience and knowledge accumulated over more than 20 years, created the Healthcare Innovation Cycle methodology that allows identifying the milestones to be achieved to reduce the risk of innovation and maximize the probabilities of success.
In 2017, CIMTI adopted this methodology to promote innovative projects in the health and social field at the Catalan level, taking into account all the particularities of the Catalan health and social system. The Healthcare Innovation Cycle is a methodology that allows innovative people and entities to advance more quickly and efficiently.
The methodology establishes a sequence of milestones from the identification of the clinical need to becoming a standard of care. To maximize the efficiency and probability of success of the project, it is important to advance in parallel in 4 different domains: clinical, market/business, regulatory and technology; in each of the milestones of the Cycle.
The 10 milestones of the Healthcare Innovation Cycle are described below:
- Need: Details about the unmet clinical need and the solutions available in the market.
- Idea: Description of the potential solution to an unresolved need.
- Proof of concept: Concepts and key components validated in models, and definition of the value proposition.
- Proof of feasibility: Demonstration of feasibility of the potential solution contrasted in models and with stakeholders.
- Proof of value: Demonstration of the solution’s potential to add value to stakeholders.
- Initial (clinical) trials: Regulated production of prototypes and collection of clinical and economic data.
- Validation of the solution: Validation of the effectiveness of the solution and the value it provides to stakeholders.
- Approval and launch: Institutional and regulatory approval and release to the market.
- Clinical use: The solution is successfully used in daily clinical practice.
- Standard of care: The solution is recognized as a standard of care.
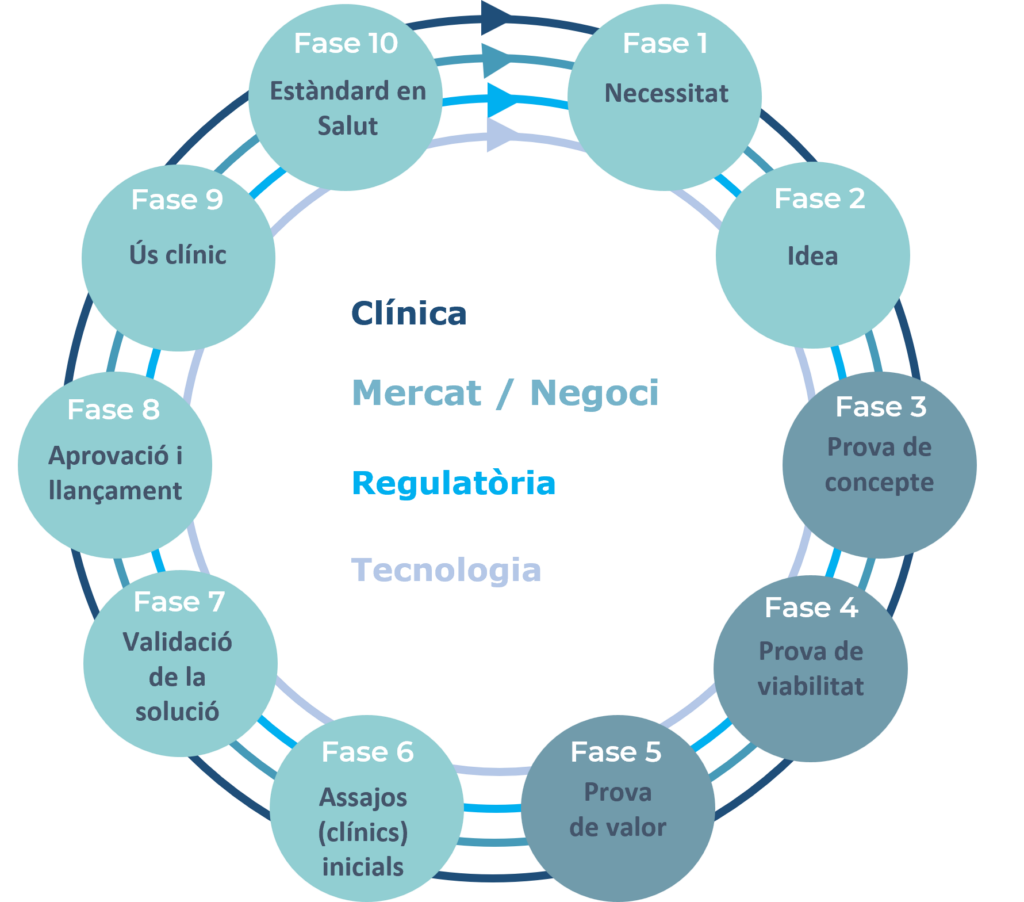
The Healthcare Innovation Cycle reduces the risk of not reaching the market that innovative solutions have. In each of the milestones, apart from the state of development of the technology, the clinical, market and business and regulatory domains are also taken into consideration. This forces us to consider the different aspects that influence innovations in health and to identify and mitigate risks. For example, apart from the aspects related to technology, it also forces us to consider whether in the clinical field, the innovation will be accepted and fits with the workflow; Or in the market and business level, if the identified unmet need has a set of potential buyers interested in acquiring the innovation at a defined price; or how many economic resources and time will have to be invested to get regulatory approval.
Finalization of support and CIMTI Alumni community
Thanks to having been selected by CIMTI, when our support ends you will be part of the CIMTI Alumni community. Being part of this community not only recognizes your time at CIMTI, but it will offer you advantages such as:
- Promotion of future collaborations in those situations where synergies exist.
- Access to private events organized by CIMTI or by any of its collaborators.
- Opportunity to participate as speakers in events organized by CIMTI.
- Appearance of your project in the CIMTI blog.
- Access to occasional support from CIMTI, at your request and subsequent evaluation by the CIMTI team.

 Català
Català Español
Español